Product Overview
The KRIBIOLISA™ Pembrolizumab ELISA Kit (Cat. No. KBI1084) is a high-sensitivity sandwich immunoassay for the quantitative determination of free, active pembrolizumab (Keytruda®) in human serum and plasma. Validated in accordance with FDA/ICH ICH Q2(R1) bioanalytical method validation guidelines, the kit employs a paratope-specific, inhibitory anti-idiotypic antibody strategy that selectively recognises the intact humanised IgG4 anti-PD-1 therapeutic antibody — distinguishing it from endogenous immunoglobulins, PD-1/PD-L1 ligands, and unrelated therapeutic monoclonal antibodies.
Pembrolizumab (Keytruda®, Merck) is a humanised monoclonal IgG4 antibody targeting programmed death-1 (PD-1), a key inhibitory receptor expressed on activated T-cells, B-cells, and natural killer (NK) cells. By blocking PD-1’s interaction with its ligands PD-L1 and PD-L2 — molecules frequently overexpressed within the tumour microenvironment — pembrolizumab reverses tumour-induced immune suppression, restoring cytotoxic T-cell activity and enabling durable antitumour immune responses. First approved by the FDA in September 2014 for unresectable or metastatic melanoma, pembrolizumab’s indications have since expanded to encompass a broad range of solid and haematological malignancies, including non-small cell lung cancer (NSCLC), head and neck squamous cell carcinoma (HNSCC), urothelial carcinoma, Hodgkin lymphoma, colorectal cancer (MSI-H/dMMR), gastric/gastroesophageal junction cancer, cervical cancer, and tumour-agnostic approvals for microsatellite instability–high (MSI-H) or mismatch repair–deficient (dMMR) solid tumours. It is listed on the World Health Organization’s (WHO) List of Essential Medicines.
As pembrolizumab biosimilar programmes advance globally and combination immuno-oncology regimens multiply, the need for precise, validated bioanalytical measurement of free circulating pembrolizumab levels has become critical for pharmacokinetic profiling, therapeutic drug monitoring, dose optimisation, and clinical response correlation. The KRIBIOLISA™ Pembrolizumab ELISA provides researchers with a robust, well-characterised tool purpose-built for these applications, with a validated dilution range
up to 64,000-fold and an accelerated stability study confirming 1-year shelf life under recommended storage conditions.
Assay Principle
The KRIBIOLISA™ Pembrolizumab ELISA uses a quantitative sandwich immunoassay format — distinct from competitive ELISA formats used in small-molecule drug assays. Human anti-pembrolizumab capture antibodies (paratope-specific, inhibitory anti-idiotypic) are pre-coated onto 96-well microplate wells. When patient samples or standards are added, free pembrolizumab in the sample binds specifically to the immobilised capture antibody. After washing to remove unbound components, an HRP-conjugated anti pembrolizumab detection antibody is added, forming a specific antibody–antigen–antibody sandwich complex. Excess conjugate is removed by washing, TMB substrate is added producing a colorimetric reaction, and the signal is stopped and read at OD450 nm.
Critically, the signal is directly proportional to pembrolizumab concentration — the higher the pembrolizumab level, the higher the OD. This is the inverse relationship to competitive ELISAs used for small-molecule drugs such as semaglutide or tirzepatide, and reflects the sandwich format standard for large therapeutic antibody quantification.
The anti-idiotypic capture antibody specifically recognises free pembrolizumab but does not recognise pembrolizumab complexed with recombinant human PD-1, nor free recombinant PD-1 itself — meaning the assay measures only the pharmacologically active, unbound (free) drug pool. This is a critical advantage for pharmacokinetic and PD-1 receptor occupancy correlative studies.
What Makes This a Sandwich ELISA — and Why It Matters for Antibody Drug Quantification
For researchers moving from small-molecule drug ELISAs (semaglutide, tirzepatide) to therapeutic antibody quantification, it is important to understand the format difference. Pembrolizumab is a large monoclonal antibody (~148 kDa) with defined epitopes, making it well-suited to a sandwich capture format. The sandwich approach for pembrolizumab offers several research advantages over competitive formats:
Sensitivity — The direct signal relationship enables detection down to LOD 2.74 ng/mL, far below clinically relevant trough concentrations, supporting studies of drug clearance and minimum effective exposure thresholds.
Specificity for free drug — The use of an anti-idiotypic capture antibody targeting the paratope (antigen-binding site) of pembrolizumab means only non-PD-1-bound drug is captured, providing true free-drug pharmacokinetic data rather than total drug levels.
Dynamic range — The 0–640 ng/mL range, combined with 64,000-fold validated dilution, covers the full spectrum from trough levels in low-dose protocols through to peak exposure in high-dose and combination regimens.
Pharmacokinetic Relevance & Recommended ELISA Ranges
Pembrolizumab is administered as a flat dose of 200 mg every 3 weeks, or 400 mg every 6 weeks, intravenously. Serum concentrations vary significantly between patients depending on body weight, immune status, tumour burden, and combination regimen. The KRIBIOLISA™ Pembrolizumab ELISA is designed to cover the full clinically encountered concentration range:
Application Expected Pembrolizumab Range (ng/mL) Recommended Working Range (ng/mL)

Assay sensitivity of <5 ng/mL is recommended for baseline pembrolizumab detection prior to treatment initiation or at extended trough time points. An upper quantification limit of ≥250 ng/mL is advised for therapeutic monitoring and high-exposure assessment in oncology settings. For samples from patients at high-dose or combination regimens exceeding the assay range, the validated 64,000 fold dilution linearity supports accurate back-calculation after dilution.
The IC₅₀ of ~216.1 ng/mL falls well within the standard therapeutic dose range (50–150 ng/mL at trough, up to ~300 ng/mL at peak), confirming the assay is appropriately sensitive across the most clinically meaningful concentration window.
Validated Performance Data
Sensitivity
- LOD: 2.74 ng/mL (mean blank + 3 SD; signal-to-noise ≥ 3:1)
- LOQ: 8.31 ng/mL (mean blank + 10 SD; signal-to-noise ≥ 10:1)
- IC₅₀: ~216.1 ng/mL
Linearity & Range
Standard (ng/mL) Mean Absorbance Interpolated (ng/mL) % Recovery
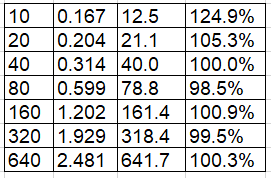
Standard curve fit: 2nd order polynomial, R² = 99.22%
Precision & Reproducibility
Assessed at three concentration levels in triplicate across three independent assay runs:
Concentration (ng/mL) Intra-Assay CV Inter-Assay CV
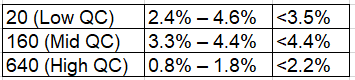
All values substantially below the FDA/ICH acceptance threshold of ≤15% CV.
Parallelism
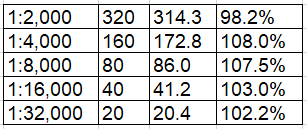
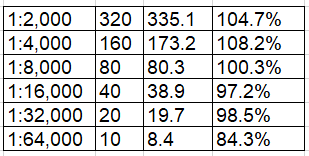
High-concentration samples were serially diluted in both human serum (1:2,000 to 1:64,000) and human plasma (1:2,000 to 1:64,000) and compared against the standard curve. Parallelism was maintained across 1:2,000 to 1:32,000 dilutions in both matrices, with % recovery within 80–120% acceptance range. The 1:64,000 dilution showed slightly reduced recovery in serum (47.1%), which is noted; at this extreme dilution, samples approach the LOD boundary. For routine laboratory use, dilutions of 1:2,000 to 1:32,000 are fully validated.
Human Serum Parallelism:
Dilution Expected (ng/mL) Interpolated (ng/mL) % Recovery
Human Plasma Parallelism:
Dilution Expected (ng/mL) Interpolated (ng/mL) % Recovery
Matrix Effect
Assay performance was compared across three conditions: buffer only, buffer
- 1:1,000 human serum, and buffer + 1:1,000 human plasma. Across the full 0–640 ng/mL standard curve range, %CV values were consistently low in both Minimal absorbance shifts were observed, confirming the absence of significant matrix interference from serum proteins, immunoglobulins, cytokines, heterophilic antibodies, or Fc-binding serum factors that are common sources of interference in therapeutic antibody ELISAs.
Accelerated Stability Study
A formal accelerated stability study was conducted by exposing kit components to 37°C stress conditions, with real-time equivalence estimated as follows:
Stress Day (37°C) Real-Time Equivalent

%CV across all standard levels remained below 15% at all time points through Day 14 (the 1-year equivalent), supporting the kit’s assigned 12-month shelf life under recommended storage conditions (2–8°C). This is a critical validation parameter for laboratories procuring kits for longitudinal study programmes.
Specificity & Selectivity
The KRIBIOLISA™ Pembrolizumab ELISA employs a paratope-specific, inhibitory anti-idiotypic capture antibody with an intrinsic affinity of KD = 0.6 nM (monovalent, measured by real-time, label-free molecular interaction analysis on immobilised pembrolizumab). This antibody targets unique idiotype or framework-region epitopes specific to pembrolizumab’s antigen-binding site, providing exceptional molecular discrimination.
Cross-reactivity profile:
Molecule Cross-Reactivity
Pembrolizumab (free) Primary analyte — fully detected
Pembrolizumab + PD-1 complex Not detected — assay is free-drug specific
Endogenous human IgG (all subclasses) Low reactivity
Nivolumab (anti-PD-1) Low reactivity
Adalimumab Low reactivity
Trastuzumab Low reactivity
Infliximab Low reactivity
Panitumumab Low reactivity
Recombinant human PD-1 Not detected
Soluble PD-L1 / PD-L2 No interference
Endogenous cytokines / matrix proteins No significant interference
The selectivity profile is particularly valuable in immuno-oncology settings where patients may have elevated endogenous immunoglobulins, receive combination regimens with other therapeutic antibodies such as nivolumab, or have circulating soluble PD-1 released from tumour-infiltrating lymphocytes.
The free-drug specificity of the capture antibody is an especially important feature: because it does not capture pembrolizumab bound to PD-1, it reports the pharmacologically available drug fraction rather than total drug — making it directly relevant to PD-1 receptor occupancy studies and exposure-response correlation analyses.
Applications
Pharmacokinetic (PK) Studies — Quantify free circulating pembrolizumab concentrations across dosing intervals (200 mg q3w or 400 mg q6w) to characterise Cmax, Cmin (trough), AUC, and clearance parameters in clinical and pre-clinical research. Validated dilution range up to 64,000-fold supports samples from peak Cmax through late trough.
Therapeutic Drug Monitoring (TDM) — Monitor individual patient pembrolizumab exposure to support personalised dosing strategies, particularly in patients with variable antibody clearance due to altered immune status, body composition, renal or hepatic comorbidities, or tumour-driven immune activation.
PD-1 Receptor Occupancy Studies — The free-drug specificity of the assay (not detecting PD-1-bound pembrolizumab) makes it an ideal tool for correlating free drug levels with PD-1 receptor saturation on circulating T-cells, supporting mechanistic immunological research.
Exposure-Response & Pharmacodynamic Correlation — Correlate quantified pembrolizumab concentrations with clinical endpoints including tumour regression, progression-free survival, T-cell activation markers, cytokine profiles, and immune-related adverse event (irAE) incidence.
Immunogenicity & Anti-Drug Antibody (ADA) Research — Use as the drug tolerance bridge in immunogenicity assays, helping establish the drug concentration at which ADA signal is detectable, a critical parameter in drug-tolerant ADA ELISA design.
Biosimilar Development & Characterisation — With multiple pembrolizumab biosimilar candidates in development globally (particularly in Asia and Europe), this kit provides a validated immunoreactivity comparability assay for biosimilar characterisation against the Keytruda® reference product.
Combination Immunotherapy Research — Measure pembrolizumab exposure in the context of combination regimens (pembrolizumab + chemotherapy, pembrolizumab + targeted agents, pembrolizumab + other checkpoint inhibitors) to evaluate whether co-administration alters pembrolizumab PK.
Tumour Biology & Immune Microenvironment Research — Quantify pembrolizumab penetration into tumour-conditioned media, cell culture supernatants, or tissue eluates to study drug distribution within the tumour immune microenvironment.
Clinical Trial Sample Analysis — Process serum and plasma samples from clinical trials investigating pembrolizumab monotherapy or combination regimens across all approved and investigational indications including melanoma, NSCLC, HNSCC, urothelial carcinoma, MSI-H tumours, and emerging indications.
Scientific Background: Pembrolizumab & PD-1 Checkpoint Blockade
Pembrolizumab (Keytruda®, Merck; formerly MK-3475 and lambrolizumab) is a humanised IgG4κ monoclonal antibody with a molecular weight of approximately 148 kDa. It was engineered as an IgG4 isotype specifically to minimise antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC), ensuring it modulates immune signalling without directly depleting T-cells — a critical design feature that contributes to its durable clinical efficacy and manageable safety profile.
Its mechanism is tumour-antigen–agnostic: pembrolizumab works by blocking the PD-1 immune checkpoint regardless of what antigen the tumour expresses, enabling broad cross-cancer application. PD-L1 expression on the tumour (measured by IHC, commonly with the 22C3 pharmDx assay) serves as a predictive biomarker for response, with tumour proportion score (TPS) or combined positive score (CPS) thresholds guiding patient selection across different indications.
Pembrolizumab’s tumour-agnostic MSI-H/dMMR approval (2017) was a landmark in oncology — the first FDA approval based on a tumour’s molecular profile rather than its anatomical origin, establishing the principle of biomarker-driven indication expansion that now underpins many immune checkpoint inhibitor development programmes.
Selected FDA approvals (non-exhaustive):
- Melanoma (unresectable or metastatic): September 2014 — first approval
- NSCLC (PD-L1 TPS ≥1%, 1L, 2L): 2015–2016
- HNSCC (recurrent/metastatic): 2016
- Hodgkin lymphoma (relapsed/refractory): 2017
- MSI-H/dMMR solid tumours (tumour-agnostic): May 2017 — first tumour-agnostic approval in oncology
- *Urothelial carcinoma, gastric/GEJ cancer, cervical cancer, hepatocellular carcinoma, Merkel cell carcinoma, TMB-H tumours, endometrial carcinoma, CRC (MSI-H), biliary tract cancer


