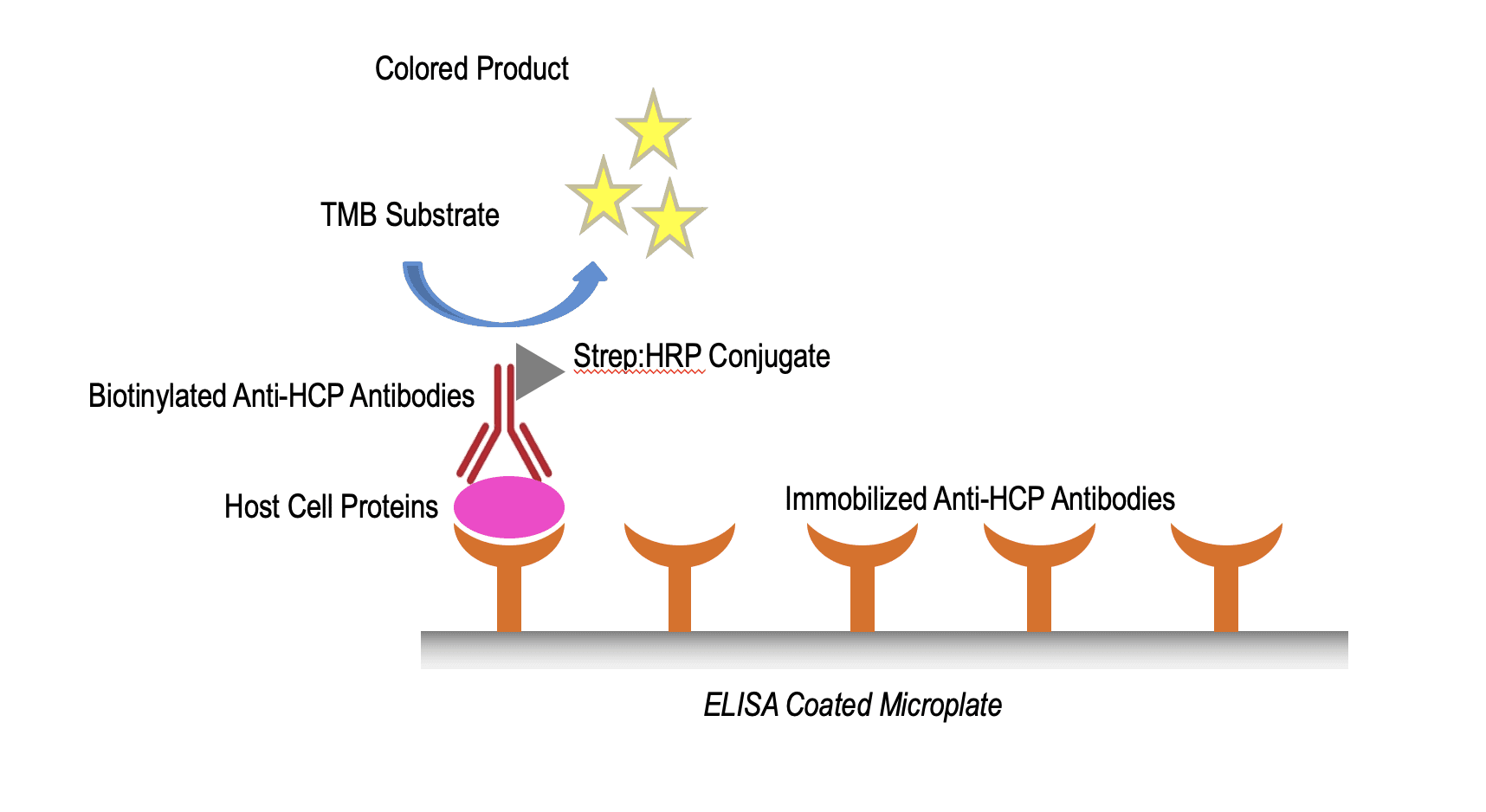
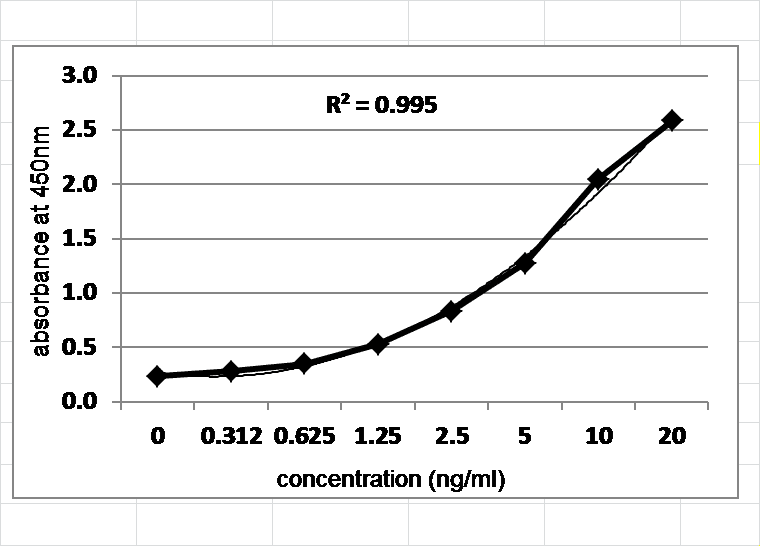
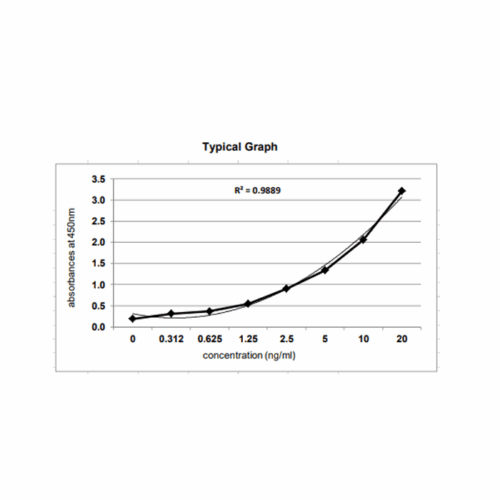
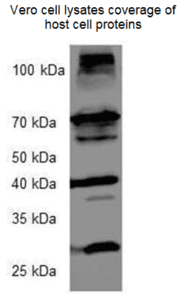
Host Cell Protein ELISA
Host Cell Protein ELISA is a critical analytical technique for biopharmaceutical and vaccine research and manufacturing. Cell lines used as expression systems inadvertently produce HCP proteins that can impact safety, efficacy, and quality of the final product. Although low levels of most contaminants may be inconsequential, patient safety importance requires that they be eliminated or reduced to the lowest levels to avoid problems such as adverse immune reactions. Impurities can have significant cost implications in drug development and manufacture. Failure to identify and remove these impurities early in drug development may lead to reduced drug efficacy or adverse patient reactions that could delay release or kill a promising drug candidate. Reducing contaminates to very low levels necessitate multiple purification steps that reduce product yield.
To ensure the safety of recombinant-expressed biopharmaceuticals as well as vaccines, health authorities are demanding the close monitoring of HCP impurities by applying HCP detection methods with high sensitivity and specificity.
By integrating HCP kits into their biologics manufacturing process, researchers can monitor contamination more effectively, identify and eliminate the root cause of contamination, and adhere to regulatory requirements. Moreover, HCP kits lower the risk of adverse events due to HCP impurity in patients, decrease the risk of regulatory non-compliance, and minimize the number of lots rejected due to high HCP levels.
The KRIBIOLISA Telescopic HCP ELISA approach for CHO, E. coli, Vero and HEK. While traditional generic HCP assays provide just a single antibody to capture the HCPs, KRISHGEN uses double antibody coverage to ensure a wider coverage of all HCPs in the band from smaller to larger sizes.
Thus, the enhanced telescopic angle coverage HCP approach results in host cell protein (HCP) assays that are demonstrably superior to traditional generic HCP assays by minimizing well-known limitations. This includes the possibility that single-kit anti-HCP antibodies might not comprehensively detect HCPs present in a manufacturer’s sample.
The manufacturing of each Telescopic generic HCP ELISA kit type involves the following steps:
– Immunization of rabbits using specific antigen material
– Isolation of polyclonal antibodies and purification
– Development of ready-to-use ELISA kits
The antibodies of each individual kit type will display specific reactivity towards a subset of the total protein profile and will show weak or no reactivity towards other proteins of given strain/cell line.
The proportion of proteins being recognized compared to the total protein is termed coverage.
- Calibrators / standards used in the kit are validated for HCP coverage.
- Antibodies used in the kit have been tested for reactivity with more than 50 proteins by Western Blot. Western Blot analysis.
- The kits have been validated successfully for testing of final and in process product HCPs in variety of products regardless of growth and purification process.
- Produced in an ISO 13485 certified facility
 KRISHGEN’s KRIBIOLISA Telescopic HCP ELISA range for Cho, E.coli, Vero, HEK has a specific advantage of using double capture antibodies to be able to offer wider detection of the proteins. Moreover, using a biotin-streptavidin detection capabilities offers a more sensitive assay compared to existing direct HRP conjugated detection antibodies in ELISAs.
KRISHGEN’s KRIBIOLISA Telescopic HCP ELISA range for Cho, E.coli, Vero, HEK has a specific advantage of using double capture antibodies to be able to offer wider detection of the proteins. Moreover, using a biotin-streptavidin detection capabilities offers a more sensitive assay compared to existing direct HRP conjugated detection antibodies in ELISAs.
Our TELESCOPIC GENERIC HCP assays offer clear distinct advantages in terms of sensitivity and specificity –
1) Critical HCP ELISA performance criteria
2) Enhanced HCP coverage
3) Sufficient HCP log-reduction
4) ELISA accuracy
5) Sample dilution linearity
- Pre-coated break-apart 96 well plates with a simple protocol
- Ready to ship (3-day lead time)
- Standards lyophilized for better stability.
Sensitivity: The limit of detection is defined as the lowest detectable concentration corresponding to a signal of Mean of ‘0’ standard plus 2* SD. Each kit mentions sensitivity in the datasheet.
Calibration: The Standards provided in the kit are calibrated against wide coverage of the host cell proteins which were validated with 2D gels. Comparator studies with other commercially available generic kits have also been done to check the recoveries obtained. Connect with us to obtain more papers on the same.
Recovery and Matrix Effect: The kits have been optimized at specific dilution to ensure optimal noise:signal ratios. Please do not change the dilution of samples recommended in the IFUs. For any queries, please connect with us directly.
TELESCOPIC HCP ASSAYS
Krishgen’s range of Host Cell Protein ELISA for measuring process samples is the telescopic generic HCP ELISA which offers 360º coverage. We offer quality HCP assays with affordable costs.
Other Available Contamination Assays from Krishgen:
Related Blogs
Featured Citations
Precision: Precision is defined as the percent coefficient of variation (%CV) i.e. standard deviation divided by the mean and multiplied by 100. Assay precision was determined by both intra and inter assay reproducibility on two pools with low, medium, and high concentrations. While actual precision may vary from laboratory to laboratory and technician to technician, it is recommended that all operators achieve precision below these design goals before reporting results.
Featured Products
Host Cell Protein ELISA
 The production process of both mAbs and viral vectors (used in gene therapy and vaccine development) involves the use of host cells, such as Chinese Hamster Ovary (CHO) cells and HEK293 or insect cells, which are genetically engineered to produce the desired antibody.
The production process of both mAbs and viral vectors (used in gene therapy and vaccine development) involves the use of host cells, such as Chinese Hamster Ovary (CHO) cells and HEK293 or insect cells, which are genetically engineered to produce the desired antibody.
During production, the host cells produce not only the desired antibody but also a multitude of HCPs. HCPs are generated as byproducts of the cellular machinery and metabolic processes of the host cells. These HCPs can vary in composition and abundance depending on several factors, including the host cell line, culture conditions, and purification methods employed. Additionally, culture conditions, including the growth medium, temperature, and oxygen levels, can affect the metabolic activities of the host cells and consequently impact HCP generation. The stage of the production process, such as cell culture, virus propagation, and purification, can also influence HCP levels.
The presence of HCPs in mAb or viral vector products can have significant impacts on both the quality and safety of the final drug product.
In mAbs: HCPs can affect the quality of mAbs by potentially altering their stability, efficacy, and immunogenicity. Even at low concentrations, certain HCPs can bind to and modify the structure of mAbs, leading to changes in their functional properties. This can result in reduced potency or altered pharmacokinetics, impacting the therapeutic effect of the drug.
In Viral Vectors: HCPs can affect the quality of the viral vector by potentially interfering with its transduction efficiency or stability. HCP impurities can reduce the viral vector’s ability to effectively deliver genetic material to target cells, resulting in reduced gene transfer efficacy. Moreover, HCPs can impact the storage stability of viral vectors, leading to a decrease in their potency over time.
Furthermore, the presence of HCPs in mAb products raises safety concerns. HCPs can elicit immune responses in patients, potentially leading to allergic reactions or other adverse events. Additionally, HCPs can interfere with the accurate measurement of product concentration and impurity profiles, complicating the quality control process.
If you are unable to locate your desired ELISA kit on our website, or have a technical query, please drop us a message and our team will come back to you with availability. If we do not have it available, we will suggest a feasible custom development option.